Industrial Gold Plating Overview
Industrial gold plating is utilized across a range of industries for both the conductive and noble properties that gold provides. Gold is the third most conductive metal behind silver and copper but unlike these metals, gold’s contact resistance is extremely consistent since gold does not oxidize or tarnish. This makes gold an excellent choice for low-voltage applications such as signal transmission when small changes in resistance can be problematic. In addition, gold provides excellent barrier corrosion resistance in many industrial applications since it does not corrode.
Functional industrial gold plating is plated as soft gold with 99.9% purity (Type III) as well as hardened 99.0-99.7% pure (Type I or II) deposits alloyed with nickel or cobalt. Unlike decorative gold applications which often use a gold flash of less than 0.00001” inches (0.25um), industrial gold deposits are normally thicker to provide improved function and durability. Industrial gold is commonly plated in thickness typically ranging between 0.00001 inches (0.25 micrometers) to 0.0001 inches (2.5 micrometers) and is commonly plated over underlayers of nickel and/or copper.
Table 1: Common Gold Plating Thickness for Functional Gold Use
| Common Thickness of Gold | Relevant ASTM B488 Class | Relevant MIL-G-45204 Class | Applications |
| 10µin
0.25µin |
Class 0.25 | N/A | Suitable for static connections in controlled environments without cyclical use. Good for solderability and wire bonding (10-20µin) |
| 30µin
0.75µin |
Class 0.75 | Class 0 | Good for connections that may be soldered. Contacts can be exposed to moderate environments and wear cycles but not high cycle or chemical attack. |
| 50µin
1.25µin |
Class 1.25 | Class 1 | Suitable for connections that may be soldered, exposed to more aggressive environments, offers superior protection against wear in moderate to high cycles. |
| 100µin
2.5µin |
Class 2.5 | Class 2 | Not recommended for connections that may be soldered, offers excellent protection against corrosive environments and durability high wear applications |
Considerations when Specifying Industrial Gold Plating – Gold Purity and Hardness
Industrial gold plating is divided into two primary categories – hard and soft gold plating – based on the hardness of the deposit. Soft gold is the highest purity (99.9% minimum) and is used where the noble properties of gold take priority over wear resistance. Soft gold lives up to its namesake with a maximum hardness of 90 Knoop; whereas hard gold plating has reduced purity (99.0-99.7% Pure) but can have hardnesses of up to 200 Knoop due to the addition of alloying elements such as nickel and cobalt. Common industrial uses for soft gold include wire bonding or soldering of electronics, lapping electrical contacts with low contact pressures (< 10 Pa), infrared reflectors and high corrosion resistance contacts or electrodes.
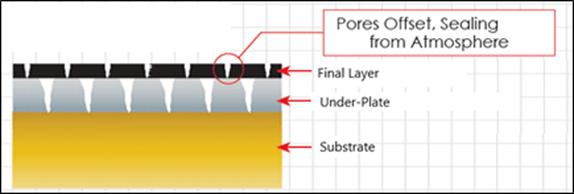
Soft gold is deposited without any alloying elements, allowing for the natural grain structure of gold, alongside gold’s natural low porosity, to provide a corrosion resistant barrier coating over the substrate. Soft gold is often seen in connector applications that are designed to remain connected for long periods of time. Soft Gold is also an excellent reflector of near-infrared (NIR) and infrared (IR) radiation and making it an excellent choice for thermal shielding in extremely low temperature quantum computing applications. The properties of soft and hard gold can also be combined into a “duplex” coating, where the hard gold outer layer provides wear resistance, while the soft gold underplate increases the corrosion resistance considerably due to the offsetting of pores between the hard and soft gold plating layers.
Hard gold is not truly hard in terms of other metals such as nickel or titanium; however, its hardness is relative compared to the very soft nature of pure gold. The increase in hardness of hard gold affords greatly improved wear resistance where sliding wear or repeated contact can abrade gold from the surface. Hard gold is commonly used in electrical terminals and contacts where higher contact pressures (> 10 Pa) and repeated engagement occurs such as male/female connectors or pogo pin contacts. In addition, hard gold plating provides a lubricious surface that is not prone to fretting and resists galling. Hard gold can be utilized for joining applications such as soldering even with a mild rosin-only flux; however hard gold is not recommended for wire bonding due to its alloying elements.
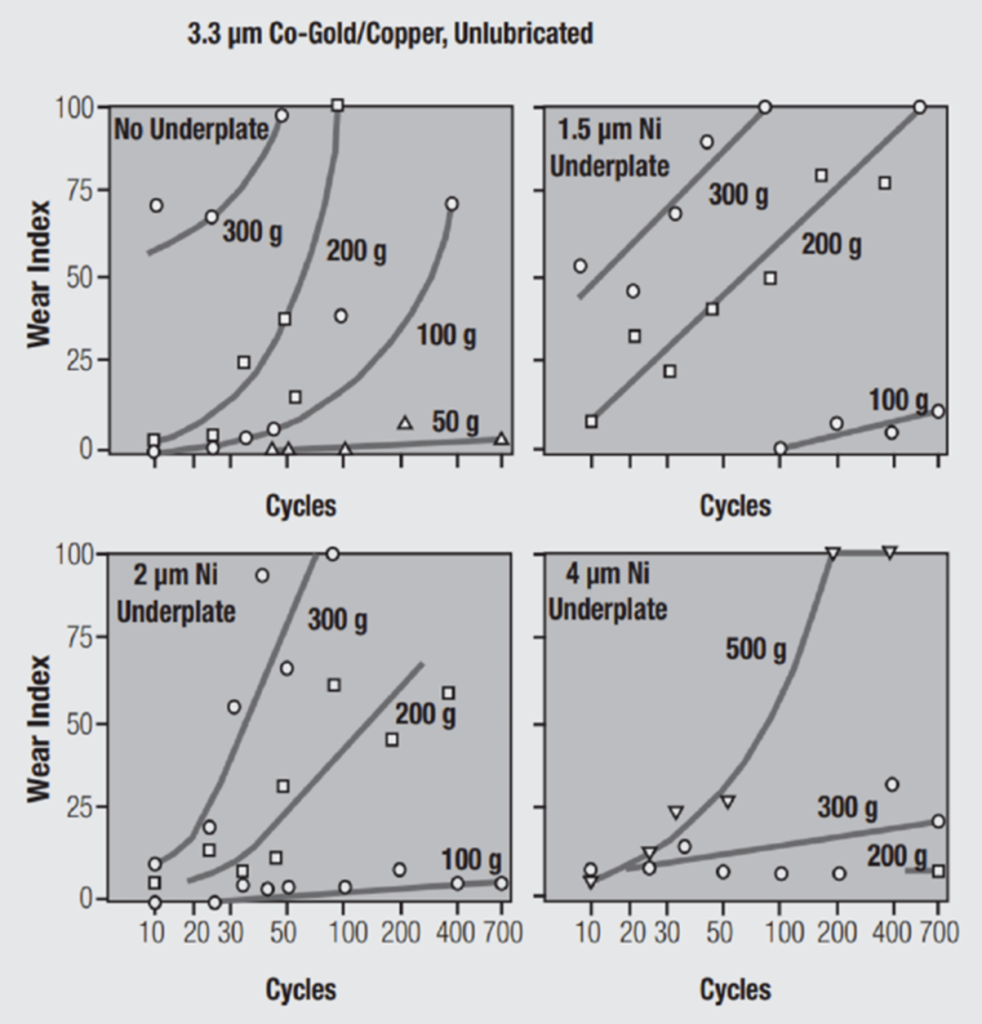
Figure 2: Impact of Nickel Underplate on Hard Gold for Wear Protection at Different Contact Pressures and Underplate Thicknesses
Comparison of Industrial Gold Plating to Other Conductive Finishes
In industrial uses, gold plating is often compared to other plated conductive finishes, most commonly, silver, copper and tin. Silver plating, like gold, is often used in connector applications. Like gold, industrial silver plating provides excellent conductivity and lubricity – even at high temperatures. However, silver readily reacts with sulfur forming a blue or black tarnish of silver sulfide. Although silver sulfide is relatively conductive, it still will increase contact resistance which can be problematic in sensitive low-voltage or signal transmission applications. While numerous anti-tarnish treatments exist, they only mitigate and do not eliminate silver tarnish from forming. Due to gold’s nonreactive nature, it will never tarnish or corrode, which makes gold desirable for applications sensitive to slight changes in contact resistance.
Copper plating, like silver and gold plating, is a highly conductive metal. In fact, copper is second only to silver in conductivity. Since copper is not a precious metal, it can be plated to a much higher thickness than silver and gold without as great of a cost impact. This is very beneficial for promoting corrosion resistance when used as an underplate since copper is a relatively noble metal that provides very good barrier corrosion protection. The downside of copper as a final finish is that copper reacts readily with sulfur, oxygen or even chlorine to form copper oxide (CuO or Cu2O), copper sulfide (CuS), copper sulfate (CuSO4) or various halides. Any of these compounds have a major impact on the conductivity of copper and will increase contact resistance and create hot-spots in conductors that can lead to thermal run-away events (fires). For this reason, copper plating is typically used as a “supporting character” in conductive finish stack-ups to help promote overall conductivity and corrosion resistance with the ultimate or topcoat being an alternative finish such as gold, silver, nickel or tin.
Tin plating is often referred to as poor-man’s silver since it provides many of the similar properties as silver but just not as well. Tin has reduced conductivity and corrosion resistance over silver or gold plating, but it performs adequately for many industrial applications. Matte tin plating provides good solderability but unlike industrial gold plating, the solderability of tin degrades over time due to the formation of tin oxides. However, tin does maintain solderability longer than nickel plating and the solderability of tin can be extended with proper packaging techniques.
Unlike gold plating, tin has a low melting point and as such, the service environment should be considered; nickel is preferred for a conductive finish in higher temperature applications where a precious metal is not needed. Tin or nickel plating are most commonly used for plating of larger conductors such as bus bars and connector terminals where the application has some forgiveness for contact resistance increasing over time. By comparison industrial gold plating is used for critical conductor applications where increases in contact resistance over time pose a design concern.
There are many considerations to account for when specifying a finish for a component. The technical sales and engineering staff at Advanced Plating Technologies (APT) can help with specifying the right plating or plating stack-up for your tin or nickel plating application. APT has over 75 years of experience plating tin and nickel across a range of industries and can assist with proper test plans and packaging methods to ensure deposit properties are maintained and protected.
A member of our engineering group can be contacted at sales@advancedplatingtech.com or 414.271.8138.
Blog Authored by Zach Hatseras, Estimating Engineer; Technical Editing by Matt Lindstedt, President – Advanced Plating Technologies
References:
- Bulwith, Ronald A “Soldering to Gold- A Practical Guide” chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://advancedplatingtech.com/wp-content/uploads/2016/04/Soldering-to-Gold-A-Practical-Guide.pdf
- Dr. Bob Mroczkowski Founder at connNtext associates Dr. Bob Mroczkowski was one of the connector world’s most significant innovators and educators. “Connector Degradation Mechanisms-Corrosion Part II.” Connector and Cable Assembly Supplier, 31 Oct. 2019, connectorsupplier.com/connector-degradation-mechanisms-corrosion-part-ii/.
- Scardinio, Dominic. “How to Prevent Corrosion of Gold Plated Contacts or Terminals.” Advanced Plating Technologies, 21 July 2023, advancedplatingtech.com/blog/prevent-corrosion-of-gold-plated-contacts-terminals/.
- Zednicek, Antonin. “Nickel Underplates and Noble Metal Finish Wear.” Passive Components Blog, 7 Sept. 2021, passive-components.eu/nickel-underplates-and-noble-metal-finish-wear/.














 smaller components to meet design requirements. In turn, the metal finishing supplier needs to be able process increasing smaller parts when processing these components. The technique of gold plating delicate and micro parts involves more than just the gold plating process. The metal finisher needs to be able to clean, rinse, dry and inspect the deposits on these difficult components as well. As such, the job shop needs to utilize not only the proper process method but have adequate line and inspection equipment to properly plate delicate and micro parts in gold.
smaller components to meet design requirements. In turn, the metal finishing supplier needs to be able process increasing smaller parts when processing these components. The technique of gold plating delicate and micro parts involves more than just the gold plating process. The metal finisher needs to be able to clean, rinse, dry and inspect the deposits on these difficult components as well. As such, the job shop needs to utilize not only the proper process method but have adequate line and inspection equipment to properly plate delicate and micro parts in gold.